"SFDA": 12 Reasons to Avoid Buying Medical Devices and Products from the Internet
2016-04-18
SFDA warned from the purchase of medical devices and products via the Internet, in order for the health and safety of users.
SFDA identified 12 reasons to avoid buying medical device or product via the Internet which they are:
- The medical device or product may not fulfill the requirements of SFDA that concerned with the safety and quality of medical products and devices that is in Saudi Arabia markets .
- It may not be manufactured under good manufacturing controls, which is under the requirements of international standards for safety and quality.
- It may not be manufactured with quality or materials suitable for human use.
- Difficulty to obtain guarantees and after-sales service, as you may miss the guidance by the tariff card on the medical device or product.
- The possibility of obtaining false or inaccurate readings as a result of incorrect calibration.
- May not be compatible with the national standards for electric energy (electric voltage: 220 volts, frequency: 60 Hz).
- The possibility of damage, caused by handling, transporting and storage in wrong ways.
- The end of the expiration date of the product.
- Was the subject of a safety warning because of manufacturing defect or problem in its safety and integrity, which requires corrective action before re-use.
- It may be a fake version of the original brand with the same name of the device, and its difficult to identify them only by specialists.
- It may carry an identifying card of other device or medical product and different batch number.
- The possibility of non-validity of therapeutic or diagnostic medical claims and allegations.
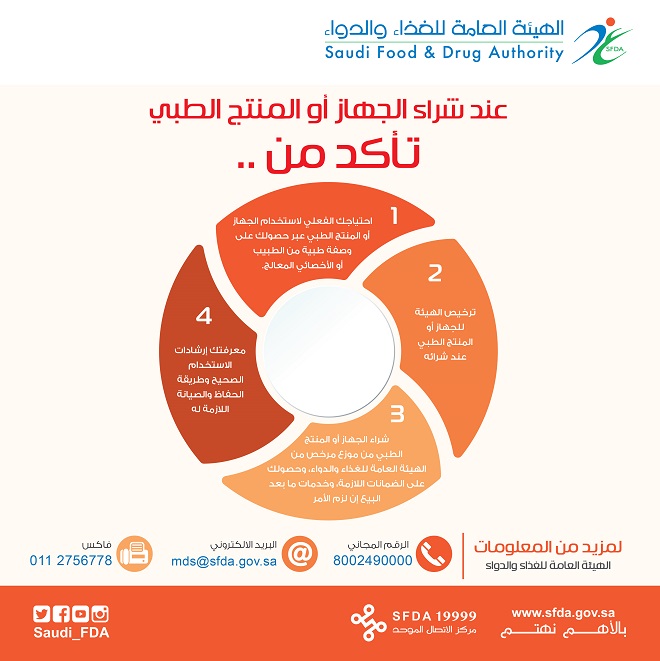
SFDA called consumers when buying a product or medical device to:
- Make sure for requiring the device or the medical product through obtaining a prescription from a doctor or specialist physician.
- Make sure the device or medical product license by SFDA when you buy it.
- Make sure you buy a product that is licensed SFDA distributor, and you have all the necessary guarantees and after-sales service, if necessary.
- Make sure that you have the correct knowledge and guidelines for using the product and the method of maintaining it, if it necessary required maintenance.
And also you can communicate with SFDA for more information, through the unified number: 19999, or by e-mail: , or by fax: 011 2756778